 Each metal atom adds one or more electrons to the sea of mobile electrons. These electrons are mobile and are distributed evenly throughout the crystal. Metallic Solids: Metals are a well-organized collection of positive ions that are surrounded and held together by a sea of free electrons.When the ionic solid is molten or dissolved in water, the ions become free to move and conduct electricity. Because the ions cannot move freely, they are electrical insulators in the solid state. Their melting and boiling points are both very high. These solids are naturally hard and brittle. Ionic solids are three-dimensional arrangements of cations and anions held together by strong electrostatic forces. Ionic Solids: Ions are the particles that make up ionic solids.Strong hydrogen bonding holds molecules of solids like H 2O together (Ice). Hydrogen-Bonded Molecular Solids – These solids’ molecules have polar covalent bonds between H and F, O, or N atoms.Examples of such solids include solid SO 2 and solid NH 3. These soft solids are electrically inactive. The molecules of these solids are held together by relatively stronger dipole-dipole interactions. Polar Molecular Solids – Polar covalent bonds form the molecules of substances such as HCl, SO 2, and others.The atoms or molecules in these solids are held together by weak dispersion forces or London forces. Non-polar Molecular Solids – These are made up of either atoms, such as argon and helium, or molecules formed by non-polar covalent bonds, such as H 2, Cl 2, and I 2.Molecular solids are further divided into three categories. Molecular Solids: In molecular solids, molecules are constituent particles.Let us now learn more about these classifications. 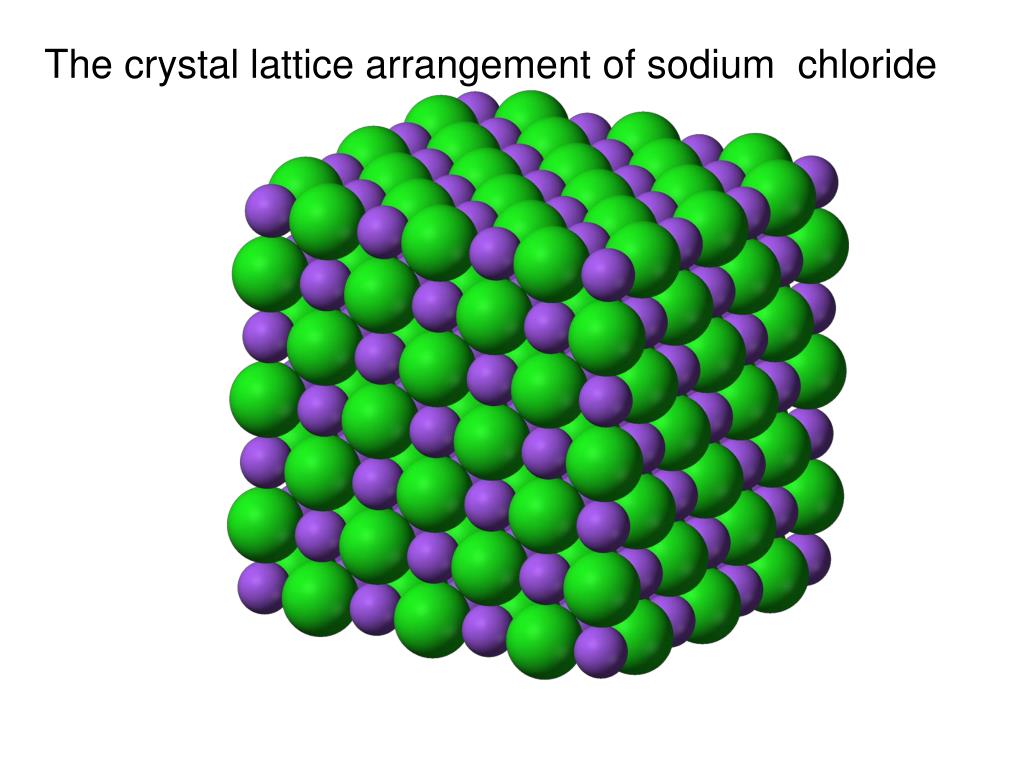
Anisotropic solids have physical properties, such as electrical resistance or refractive index, that differ when measured in different directions within the same crystal.Ĭrystalline solids are classified into four types based on the nature of their intermolecular forces: molecular, ionic, metallic, and covalent solids. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
